Virology
Prions, Viroids and Virusoids
Prions
Microorganisms
Microorganisms infecting humans and causing diseases
Protein Only Theory
Protein "infecting" humans and causing disease - Not ok
Qualifying word is organisms
What are Prions?
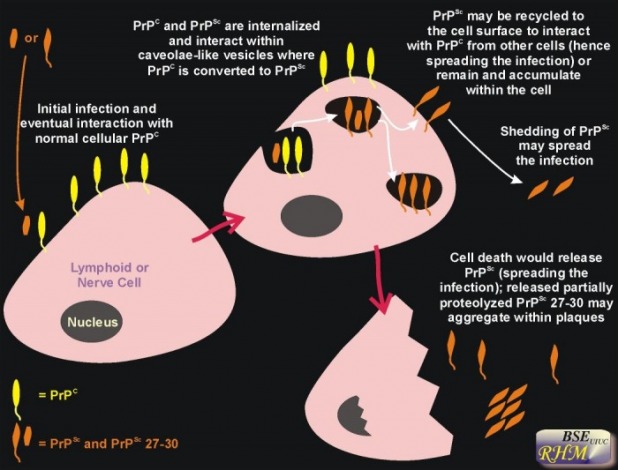
Prions are unprecedented infectious pathogens that cause a group of invariably fatal neurodegenerative diseases by an entirely novel mechanism. They are rogue protein that transform other cellular protein(PrPC) to the prion from PrPSC and are resistant to proteinase. Prion diseases may present as genetic, infectious, or sporadic disorders, all of which involve modification of the prion protein (PrP). Bovine spongiform encephalopathy (BSE), scrapie of sheep, and Creutzfeldt-Jakob disease (CJD) of humans are among the most notable prion diseases.
Prions are transmissible particles that are devoid of nucleic acid and seem to be composed exclusively of a modified protein (PrPSc). The normal, cellular PrP (PrPC) is converted into PrPSc through a posttranslational process during which it acquires a high beta-sheet content. The species of a particular prion is encoded by the sequence of the chromosomal PrP gene of the mammals in which it last replicated. In contrast to pathogens carrying a nucleic acid genome, prions appear to encipher strain-specific properties in the tertiary structure of PrPSc. While knowledge about prions has profound implications for studies of the structural plasticity of proteins, investigations of prion diseases suggest that new strategies for the prevention and treatment of these disorders may also find application in the more common degenerative diseases.
Prions are transmissible particles that are devoid of nucleic acid and seem to be composed exclusively of a modified protein (PrPSc). The normal, cellular PrP (PrPC) is converted into PrPSc through a posttranslational process during which it acquires a high beta-sheet content. The species of a particular prion is encoded by the sequence of the chromosomal PrP gene of the mammals in which it last replicated. In contrast to pathogens carrying a nucleic acid genome, prions appear to encipher strain-specific properties in the tertiary structure of PrPSc. While knowledge about prions has profound implications for studies of the structural plasticity of proteins, investigations of prion diseases suggest that new strategies for the prevention and treatment of these disorders may also find application in the more common degenerative diseases.
History of Prion Dieseases
1730s: Scrapie first appears:
- A written record is found on Scrapie in English sheep, the disease is seen all throughout
Europe. The diesease affecting sheeps and goats are called scrapie beacause the animals
scraped their fleece or skin against rocks, trees or fences. Reason being that the disease
apparently causes an itching sensation in the animals.
- A written record is found on Scrapie in English sheep, the disease is seen all throughout
Europe. The diesease affecting sheeps and goats are called scrapie beacause the animals
scraped their fleece or skin against rocks, trees or fences. Reason being that the disease
apparently causes an itching sensation in the animals.
History behind Prions
- In 1920, Hans Creutzfeldt and Alfons Maria Jakob had described the disease in man
known as Hans Creutzfeldt-Jakob disease.
- Experimental transmission of scrapie by Cuille and Chelle in 1936.
known as Hans Creutzfeldt-Jakob disease.
- Experimental transmission of scrapie by Cuille and Chelle in 1936.
Stanley Ben Prusiner
- American neurologist and biochemist at University of California, San Francisco
- Prusiner discovered prions, a class of infectious self-reproducing pathogens primarily or
solely composed of protein from "proteinaceous infections particle that lacks nucleic
acid."
- Albert Lasker Award for Basic Medical Research in 1994 and the Nobel Prize in
Physiology or Medicine in 1997 for his prion research.
- American neurologist and biochemist at University of California, San Francisco
- Prusiner discovered prions, a class of infectious self-reproducing pathogens primarily or
solely composed of protein from "proteinaceous infections particle that lacks nucleic
acid."
- Albert Lasker Award for Basic Medical Research in 1994 and the Nobel Prize in
Physiology or Medicine in 1997 for his prion research.
Different Types of Prions Viruses
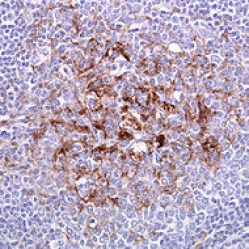
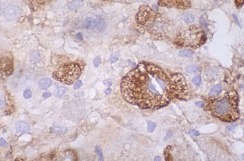
Tonsil biopsy in variant CJD. Prion Protein immunostaining.
Creutzfeldt–Jakob disease or CJD is a degenerative neurological disorder (brain disease) that is incurable and invariably fatal.It is the most common among the types of transmissible spongiform encephalopathy found in humans
Transmissible spongiform encephalopathies also known as prion diseases are a group of progressive conditions that affect the brain and nervous system of many animals, including humans.
Scrapie is a fatal, degenerative disease that affects the nervous systems of sheep and goats.It is one of several transmissible spongiform encephalopathies (TSEs), which are related to bovine spongiform encephalopathy (BSE or "mad cow disease") and chronic wasting disease of deer. Like other spongiform encephalopathies, scrapie is caused by a prion. Scrapie has been known since 1732 and does not appear to be transmissible to humans.
Transmissible spongiform encephalopathies also known as prion diseases are a group of progressive conditions that affect the brain and nervous system of many animals, including humans.
Scrapie is a fatal, degenerative disease that affects the nervous systems of sheep and goats.It is one of several transmissible spongiform encephalopathies (TSEs), which are related to bovine spongiform encephalopathy (BSE or "mad cow disease") and chronic wasting disease of deer. Like other spongiform encephalopathies, scrapie is caused by a prion. Scrapie has been known since 1732 and does not appear to be transmissible to humans.
How was scrapie transmitted to cows?
In 1970: The same symptoms appeared in cow.
Probable reason: Cows were fed meat-and-bone meal that contained scrapie infected sheep
products.
- Species to species transmission requires time.
Probable reason: Cows were fed meat-and-bone meal that contained scrapie infected sheep
products.
- Species to species transmission requires time.
The Mechanism of causing disease of Prions
1. Transform membrane glycoprotein, PrPC, to the prions form PrPSC.
2. More and more PrPC gets transformed
3. Cells work poorly or do not work at all.
4. Cells die, release prions into bloodstream to reinfect other cells.
2. More and more PrPC gets transformed
3. Cells work poorly or do not work at all.
4. Cells die, release prions into bloodstream to reinfect other cells.
PrPC
- A normal protein found on the membranes of cells.
- It has 209 amino acids(in humans), one disulfide bond and a mainly alpha-helical structure.
- Several topological forms exist as one cell surface form anchored via glycolipid and two
transmembrane forms.
- The normal protein is not sedimentable which means it cannot be separated by centrifuging
techniques.
- PrP has been reported to play important roles in cell-cell adhesion and intracellular signaling
in vivo, and may therefore be involved in cell-cell communication in the brain.
- It has 209 amino acids(in humans), one disulfide bond and a mainly alpha-helical structure.
- Several topological forms exist as one cell surface form anchored via glycolipid and two
transmembrane forms.
- The normal protein is not sedimentable which means it cannot be separated by centrifuging
techniques.
- PrP has been reported to play important roles in cell-cell adhesion and intracellular signaling
in vivo, and may therefore be involved in cell-cell communication in the brain.
PrPSC
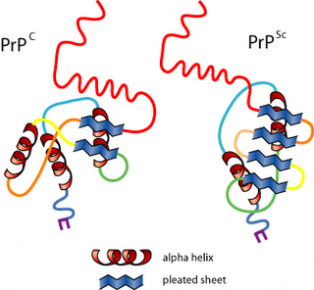
The infectious isoform of PrP, known as PrPSc, is able to convert normal PrPC proteins into the infectious isoform by changing their conformation and in turn, it alters the way the proteins interconnect. Although the exact 3D structure of PrPSc is not known, it has a higher proportion of β-sheet structure in place of the normal α-helix structure. Aggregations of these abnormal isoforms form highly structured amyloid fibers, which accumulate to form plaques. It is unclear if these aggregates are the cause of cell damage or are simply a side effect of the underlying disease process The end of each fiber acts as a template onto which free protein molecules may attach, allowing the fiber to grow. Only PrP molecules with an identical amino acid sequence to the infectious PrPSc are incorporated into the growing fiber.
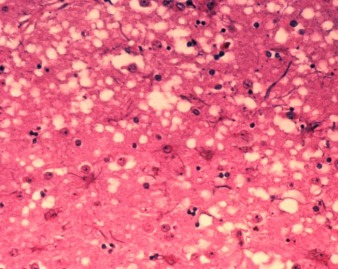
This micrograph of brain tissue reveals the cytoarchitectural histopathologic changes found in bovine spongiform encephalopathy. The presence of vacuoles, i.e. microscopic “holes” in the gray matter, gives the brain of BSE-affected cows a sponge-like appearance when tissue sections are examined in the lab.
How to transform?
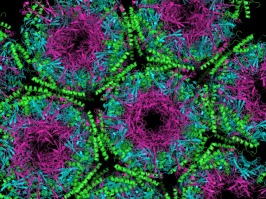
The crystallographic structure of the Prion protein
- PrPSC forms a heterodimer with normal PrPC.
- Template for altering the protein fold.
- Prion diseases are associated with the conversion
of tightly alpha-helix rich prion protein(PrPC) into
loose beta-structure-rich insoluble conformer
(PrPSc) that is thought to be infectious.
http://www.scitech.ac.uk/PMC/PRe...ion.aspx
- Template for altering the protein fold.
- Prion diseases are associated with the conversion
of tightly alpha-helix rich prion protein(PrPC) into
loose beta-structure-rich insoluble conformer
(PrPSc) that is thought to be infectious.
http://www.scitech.ac.uk/PMC/PRe...ion.aspx
Mouse with knockout PrPC gene
The mouse with knockout PrPC gene does not contain PrPC glycoprotein so when it is infected with prions, there is nothing for the prions to convert hence the mice lives.
Mouse with normal PrPC gene
The mouse with normal PrPC gene is able to produce PrPC protein, when it is infected with prions, the prions convert normal PrPC to rogue proteins hence the mice dies
Transmission

Transmission of prions includes eating contaminated beef or mutton and it can also be acquired through blood transfusion.
Viroids
Viroids - Subviral pathogens
Viroids are infectious plant pathogens that consist of a short stretch (a few hundred nucleobases) of highly complementary, circular, single-stranded RNA without the protein coat that is typical for viruses.
Mechanism of action
Viroid RNA does not code for any protein. The replication mechanism involves RNA polymerase II, an enzyme normally associated with synthesis of messenger RNA from DNA, which instead catalyzes "rolling circle" synthesis of new RNA using the viroid's RNA as template. Some viroids are ribozymes, having catalytic properties which allow self-cleavage and ligation of unit-size genomes from larger replication intermediates.
Ribozymes are RNA molecules that hydrolyze their own phodphodiester bonds, or the hydrolysis of bonds in other RNAs, but they have also been found to catalyze the aminotransferase activity of the ribosome.
Ribozymes are RNA molecules that hydrolyze their own phodphodiester bonds, or the hydrolysis of bonds in other RNAs, but they have also been found to catalyze the aminotransferase activity of the ribosome.
Disease caused by viroid
Viral Hepatitis D
The only known human disease known to be caused by a viroid-like entity is Hepatitis D. Hepaptitis D was previously ascribed to a defective virus called the delta agent. Delta agent is a type of virus called hepatitis D that causes symptoms only in people who have a hepatitis B infection. There is an extensive sequence complementarity between the hepatitis D viroid RNA and human liver cell 7S RNA. The 7S RNA structure involved in the translocation of secretory and membrane- associated particles. The hepatitis D viroid
causes liver cell death via sequestering this 7S RNA
and/or cleaving it.
causes liver cell death via sequestering this 7S RNA
and/or cleaving it.
Transmission of Hepatitis D
- Co-infection with Hepatitis B virus
- Bodily fluid such as unprotected sex, sharing of contaminated
needles and close proximity
- Bodily fluid such as unprotected sex, sharing of contaminated
needles and close proximity
Virusoid
What is a virusoid?
Virusoid is an infectious agent that infects plants in conjunction with an assistant virus.
Note: Viroids are circular RNA molecules that infects plants but do not require an assistant virus
Note: Viroids are circular RNA molecules that infects plants but do not require an assistant virus
Diagnosis of Prions Disease
- Microscopic examination of human brain tissue section
- Immunohistochemistry using prions-specific antibodies
- Latest: Conformation dependent immunoassay (CDI)
- Immunohistochemistry using prions-specific antibodies
- Latest: Conformation dependent immunoassay (CDI)